Search results
Search for "electrophilic substitution" in Full Text gives 62 result(s) in Beilstein Journal of Organic Chemistry.
Green and sustainable approaches for the Friedel–Crafts reaction between aldehydes and indoles
- Periklis X. Kolagkis,
- Eirini M. Galathri and
- Christoforos G. Kokotos
Beilstein J. Org. Chem. 2024, 20, 379–426, doi:10.3762/bjoc.20.36
- pharmaceutical and biological activities. Among indole derivatives, bis(indolyl)methanes (BIMs) are profoundly interesting, due to their wide range of pharmaceutical properties. The most common approach involves the electrophilic substitution of various aldehydes and ketones by indoles, utilizing either protic

Graphical Abstract

Scheme 1: Examples of BIMs used for their medicinal properties.

Scheme 2: Mechanisms for the synthesis of BIMs using protic or Lewis acids as catalysts.

Scheme 3: Synthesis of bis(indolyl)methanes using DBDMH.

Scheme 4: Competition experiments and synthesis of bis(indolyl)methanes using DBDMH.

Scheme 5: Proposed mechanism for formation of BIM of using DBDMH.

Scheme 6: Synthesis of bis(indolyl)methanes using I2.

Scheme 7: General reaction mechanism upon halogen bonding.

Scheme 8: Synthesis of bis(indolyl)methanes using I2, introduced by Ji.

Scheme 9: Synthesis of bis(indolyl)methanes using Br2 in CH3CN.

Scheme 10: Βidentate halogen-bond donors.

Scheme 11: Synthesis of bis(indolyl)methanes using bidentate halogen-bond donor 26.

Scheme 12: Proposed reaction mechanism.

Scheme 13: Synthesis of bis(indolyl)methanes using iodoalkyne as catalyst.

Scheme 14: Proposed reaction mechanism.

Scheme 15: Optimized reaction conditions used by Ramshini.

Scheme 16: Activation of the carbonyl group by HPA/TPI-Fe3O4.

Scheme 17: Synthesis of BIMs in the presence of nanoAg-Pt/SiO2-doped silicate.

Scheme 18: Mechanism of action proposed by Khalafi-Nezhad et al.

Scheme 19: Activation of the carbonyl group by the Cu–isatin Schiff base complex.

Scheme 20: Optimum reaction conditions published by Jain.

Scheme 21: Organocatalytic protocol utilizing nanoparticles introduced by Bankar.

Scheme 22: Activation of the carbonyl group by the AlCl3·6H2O-SDS-SiO2 complex.

Scheme 23: Optimal reaction conditions for the aforementioned nano-Fe3O4 based catalysts.

Scheme 24: Nanocatalytic protocol proposed by Kaur et al.

Scheme 25: Microwave approach introduced by Yuan.

Scheme 26: Microwave approach introduced by Zahran et al.

Scheme 27: Microwave irradiation protocol introduced by Bindu.

Scheme 28: Silica-supported microwave irradiation protocol.

Scheme 29: Proposed mechanism for formation of BIM by Nongkhlaw.

Scheme 30: Microwave-assisted synthesis of BIMs catalyzed by succinic acid.

Scheme 31: Proposed mechanism of action of MMO-4.

Scheme 32: Catalytic approach introduced by Muhammadpoor-Baltork et al.

Scheme 33: Reaction conditions used by Xiao-Ming.

Scheme 34: Ultrasonic irradiation-based protocol published by Saeednia.

Scheme 35: Pyruvic acid-mediated synthesis of BIMs proposed by Thopate.

Scheme 36: Synthesis of BIMs using [bmim]BF4 or [bmim]PF6 ionic liquids.

Scheme 37: Synthesis of BIMs utilizing In(OTf)3 in octylmethylimidazolium hexafluorophosphate as ionic liquid.

Scheme 38: FeCl3·6H2O-catalyzed synthesis of BIMs with use of ionic liquid.

Scheme 39: Synthesis of BIMs utilizing the [hmim]HSO4/EtOH catalytic system.

Scheme 40: Synthesis of BIMs utilizing acidic ionic liquid immobilized on silica gel (ILIS-SO2Cl).

Scheme 41: The [bmim][MeSO4]-catalyzed reaction of indole with various aldehydes.

Scheme 42: The role of [bmim][MeSO4] in catalyzing the reaction of indole with aldehydes.

Scheme 43: Synthesis of BIMs utilizing FeCl3-based ionic liquid ([BTBAC]Cl-FeCl3) as catalyst.

Scheme 44: Synthesis of BIMs using [Msim]Cl at room temperature.

Scheme 45: [Et3NH][H2PO4]-catalyzed synthesis of bis(indolyl)methanes.

Scheme 46: PILs-catalyzed synthesis of bis(indolyl)methanes.

Scheme 47: FSILs-mediated synthesis of bis(indolyl)methanes.

Scheme 48: Possible “release and catch” catalytic process.

Scheme 49: Synthesis of bis(indolyl)methanes by [DABCO-H][HSO4].

Scheme 50: Synthesis of bis(indolyl)methanes by [(THA)(SO4)].

Scheme 51: Synthesis of BBSI-Cl and BBSI-HSO4.

Scheme 52: Synthesis of BIMs in the presence of BBSI-Cl and BBSI-HSO4.

Scheme 53: Chemoselectivity of the present method.

Scheme 54: Synthesis of BIMs catalyzed by chitosan-supported ionic liquid.

Scheme 55: Proposed mechanism of action of CSIL.

Scheme 56: Optimization of the reaction in DESs.

Scheme 57: Synthesis of BIMs using ChCl/SnCl2 as DES.

Scheme 58: Synthesis of BIMs derivatives in presence of DES.

Scheme 59: BIMs synthesis in choline chloride/urea (CC/U).

Scheme 60: Flow chemistry-based synthesis of BIMs by Ley.

Scheme 61: Flow chemistry-based synthesis of BIMs proposed by Nam et al.

Scheme 62: Amino-catalyzed reaction of indole with propionaldehyde.

Scheme 63: Aminocatalytic synthesis of BIMs.

Scheme 64: Proposed mechanism for the aminocatalytic synthesis of BIMs.

Scheme 65: Enzymatic reaction of indole with aldehydes.

Scheme 66: Proposed mechanism for the synthesis of BIMs catalyzed by TLIM.

Scheme 67: Proposed reaction mechanism by Badsara.

Scheme 68: Mechanism proposed by D’Auria.

Scheme 69: Photoinduced thiourea catalysis.

Scheme 70: Proposed mechanism of photoacid activation.

Scheme 71: Proposed mechanism of action for CF3SO2Na.

Scheme 72: Proposed mechanism for the synthesis of BIMs by Mandawad.

Scheme 73: Proposed mechanism for the (a) acid generation and (b) synthesis of BIMs.

Scheme 74: a) Reaction conditions employed by Khaksar and b) activation of the carbonyl group by HFIP.

Scheme 75: Activation of the carbonyl group by the PPy@CH2Br through the formation of a halogen bond.

Scheme 76: Reaction conditions utilized by Mhaldar et al.

Scheme 77: a) Reaction conditions employed by López and b) activation of the carbonyl group by thiourea.

Scheme 78: Infrared irradiation approach introduced by Luna-Mora and his research group.

Scheme 79: Synthesis of BIMs with the use of the Fe–Zn BMOF.
Copper-promoted C5-selective bromination of 8-aminoquinoline amides with alkyl bromides
- Changdong Shao,
- Chen Ma,
- Li Li,
- Jingyi Liu,
- Yanan Shen,
- Chen Chen,
- Qionglin Yang,
- Tianyi Xu,
- Zhengsong Hu,
- Yuhe Kan and
- Tingting Zhang
Beilstein J. Org. Chem. 2024, 20, 155–161, doi:10.3762/bjoc.20.14
- intermediate C is then generated, followed by the combination of the bromine anion with intermediate B. Finally, selective C5 bromination is accomplished via aromatic electrophilic substitution of 1a with intermediate C promoted by the copper catalyst to afford the desired product 3aa. Conclusion In summary

Graphical Abstract

Scheme 1: Methods for the C5-selective bromination of 8-aminoquinoline amides.

Scheme 2: Substrate scope of the 8-aminoquinoline amides. Reaction conditions: 1 (0.2 mmol), 2a (0.8 mmol), C...

Scheme 3: Substrate scope of the bromoalkanes. Reaction conditions: 1a (0.2 mmol), 2 (0.8 mmol), Cu(OAc)2·H2O...

Scheme 4: Further substrate scope investigations and gram-scale application.

Scheme 5: Control experiments and proposed mechanism.
α-(Aminomethyl)acrylates as acceptors in radical–polar crossover 1,4-additions of dialkylzincs: insights into enolate formation and trapping
- Angel Palillero-Cisneros,
- Paola G. Gordillo-Guerra,
- Fernando García-Alvarez,
- Olivier Jackowski,
- Franck Ferreira,
- Fabrice Chemla,
- Joel L. Terán and
- Alejandro Perez-Luna
Beilstein J. Org. Chem. 2023, 19, 1443–1451, doi:10.3762/bjoc.19.103
- protodemetalation to provide ultimately the 1,4-addition adduct. In the presence of carbonyl acceptors, aldol condensation occurs providing overall a tandem 1,4-addition–aldol process. When a tert-butanesulfinyl moiety is present on the nitrogen atom, these electrophilic substitution reactions occur with good
- regardless of its nitrogen substitution; the outcome of the reaction is a tandem 1,4-addition–aldol process. When the tert-butanesulfinyl moiety is present on the nitrogen atom, electrophilic substitution of the intermediate enolates (protodemetalation or aldol condensation) occurs with decent levels of
- is poised to undergo β-fragmentation, but this process can be outcompeted by in situ electrophilic substitution reactions which offer synthetically useful procedures: 1,4-addition (for substrates having N–H bonds) or tandem 1,4-addition–aldol reactions (in the presence of carbonyl electrophiles

Graphical Abstract

Scheme 1: Air-promoted radical chain reaction of dialkylzinc reagents with α,β-unsaturated carbonyl compounds....

Scheme 2: Enolate formation by zinc radical transfer (SH2 on dialkylzinc reagents).

Scheme 3: Preparation of α-(aminomethyl)acrylate 10.

Scheme 4: Reaction of α-(aminomethyl)acrylate 10 with Et2Zn in the presence of air.

Scheme 5: Chemical correlation to determine the configuration of the major diastereomer of (RS)-14b.

Scheme 6: Air-promoted tandem 1,4-addition–aldol condensation reactions of Et2Zn with α-(aminomethyl)acrylate...

Scheme 7: Diagnostic experiments for a radical mechanism and for enolate formation.

Scheme 8: Diagnostic experiments with N-benzyl enoate 10.

Scheme 9: Reactivity manifolds for the air-promoted tandem 1,4-addition–electrophilic substitution reaction b...
Aromatic C–H bond functionalization through organocatalyzed asymmetric intermolecular aza-Friedel–Crafts reaction: a recent update
- Anup Biswas
Beilstein J. Org. Chem. 2023, 19, 956–981, doi:10.3762/bjoc.19.72
- -functionalization of the indole (Scheme 2) [25]. In 2018, Lin and co-workers deployed pyrroles 9 in an aza-Friedel–Crafts reaction with trifluoromethyldihydrobenzoazepinoindoles 8 to achieve the aromatic electrophilic substitution at the C2 position of the pyrrole ring. A further extension of the scope of this
- presence of an electron-donating functional group is crucial in the ring to activate it for aromatic electrophilic substitution processes. In 2019, Zhang and co-workers succeeded in the C6-selective aminoalkylation of 2,3-disubstituted indoles 4 without the presence of a directing group in the benzene ring
- ketimines 52 proceeding through C2 functionlization and follow up oxidation to provide 2-substitued indoles 56 which are typically difficult to obtain directly from unsubstituted indoles through electrophilic substitution. The process was catalyzed by the chiral phosphoric acid P17 to install a quaternary

Graphical Abstract

Scheme 1: First organocatalyzed asymmetric aza-Friedel–Crafts reaction.

Scheme 2: Aza-Friedel–Crafts reaction between indoles and cyclic ketimines.

Scheme 3: Aza-Friedel–Crafts reaction utilizing trifluoromethyldihydrobenzoazepinoindoles as electrophiles.

Scheme 4: Aza-Friedel–Crafts reaction utilizing cyclic N-sulfimines as electrophiles.

Scheme 5: Aza-Friedel–Crafts reaction involving N-unprotected imino ester as electrophile.

Scheme 6: Aza-Friedel–Crafts and lactonization cascade.

Scheme 7: One-pot oxidation and aza-Friedel–Crafts reaction.

Scheme 8: C1 and C2-symmetric phosphoric acids as catalysts.

Scheme 9: Aza-Friedel–Crafts reaction using Nps-iminophosphonates as electrophiles.

Scheme 10: Aza-Friedel–Crafts reaction between indole and α-iminophosphonate.

Scheme 11: [2.2]-Paracyclophane-derived chiral phosphoric acids as catalyst.

Scheme 12: Aza-Friedel–Crafts reaction through ring opening of sulfamidates.

Scheme 13: Isoquinoline-1,3(2H,4H)-dione scaffolds as electrophiles.

Scheme 14: Functionalization of the carbocyclic ring of substituted indoles.

Scheme 15: Aza-Friedel–Crafts reaction between unprotected imines and aza-heterocycles.

Scheme 16: Anilines and α-naphthols as potential nucleophiles.

Scheme 17: Solvent-controlled regioselective aza-Friedel–Crafts reaction.

Scheme 18: Generating central and axial chirality via aza-Friedel–Crafts reaction.

Scheme 19: Reaction between indoles and racemic 2,3-dihydroisoxazol-3-ol derivatives.

Scheme 20: Exploiting 5-aminoisoxazoles as nucleophiles.

Scheme 21: Reaction between unsubstituted indoles and 3-alkynylated 3-hydroxy-1-oxoisoindolines.

Scheme 22: Synthesis of unnatural amino acids bearing an aza-quaternary stereocenter.

Scheme 23: Atroposelective aza-Friedel–Crafts reaction.

Scheme 24: Coupling of 5-aminopyrazole and 3H-indol-3-ones.

Scheme 25: Pyrophosphoric acid-catalyzed aza-Friedel–Crafts reaction on phenols.

Scheme 26: Squaramide-assisted aza-Friedel–Crafts reaction.

Scheme 27: Thiourea-catalyzed aza-Friedel–Crafts reaction.

Scheme 28: Squaramide-catalyzed reaction between β-naphthols and benzothiazolimines.

Scheme 29: Thiourea-catalyzed reaction between β-naphthol and isatin-derived ketamine.

Scheme 30: Quinine-derived molecule as catalyst.

Scheme 31: Cinchona alkaloid as catalyst.

Scheme 32: aza-Friedel–Crafts reaction by phase transfer catalyst.

Scheme 33: Disulfonamide-catalyzed reaction.

Scheme 34: Heterogenous thiourea-catalyzed aza-Friedel–Crafts reaction.

Scheme 35: Total synthesis of (+)-gracilamine.

Scheme 36: Total synthesis of (−)-fumimycin.
Strategies in the synthesis of dibenzo[b,f]heteropines
- David I. H. Maier,
- Barend C. B. Bezuidenhoudt and
- Charlene Marais
Beilstein J. Org. Chem. 2023, 19, 700–718, doi:10.3762/bjoc.19.51

- authors postulated an intramolecular electrophilic substitution via a carbocation intermediate 42 (Scheme 9). Elliott et al. [47] investigated several methods to synthesise substituted dibenzo[b,f]azepines, which included the ring expansion of N-arylindoles 41 to synthesise 43 and the rearrangements of 9

Graphical Abstract

Figure 1: Dibenzo[b,f]azepine (1a), -oxepine (1b) and -thiepine (1c) as examples of dibenzo[b,f]heteropines (1...

Figure 2: Selected pharmaceuticals with the dibenzo[b,f]azepine skeleton.

Figure 3: Examples of 10,11-dihydrodibenzo[b,f]azepine-based ligands.

Figure 4: The dibenzo[b,f]azepine moiety in dyes with properties suitable for the use in organic light emitti...

Figure 5: Selective bioactive natural products (13–18) containing the dibenzo[b,f]oxepine scaffold and Novart...

Scheme 1: Retrosynthetic approach to 5H-dibenzo[b,f]azepine (1a) from nitrotoluene (22).

Scheme 2: Oxidative coupling of o-nitrotoluene (22) and reduction of 2,2'-dinitrobibenzyl (21) to form 2,2'-d...

Scheme 3: Synthesis of 10,11-dihydro-5H-dibenzo[b,f]azepine (2a) via amine condensation.

Scheme 4: Catalytic reduction of 10,11-dihydro-5H-dibenzo[b,f]azepine (2a).

Scheme 5: The Wagner–Meerwein rearrangement of acridin-9-ylmethanol (23) into 5H-dibenzo[b,f]azepine (1a).

Scheme 6: Oxidative ring expansion of 2-(9-xanthenyl)malonates 24.

Scheme 7: Ring expansion via C–H functionalisation.

Scheme 8: The synthesis of fluorinated 5H-dibenzo[b,f]azepine 38 from isatin (32).

Scheme 9: The synthesis of substituted dibenzo[b,f]azepines 43 from indoles 39.

Scheme 10: Retrosynthetic pathways to dibenzo[b,f]azepines via Buchwald–Hartwig amination.

Scheme 11: Synthesis of dibenzo[b,f]oxepine 54 and -azepine 55 derivatives via (i) Heck reaction and (ii) Buch...

Scheme 12: Double Buchwald–Hartwig amination and thioetherification in the synthesis of tricyclic azepines 60 ...

Scheme 13: Double Buchwald–Hartwig amination towards substituted dibenzoazepines 62.

Scheme 14: Double Buchwald–Hartwig amination towards 10,11-dihydro-5H-dibenzo[b,f]azepine derivatives 71.

Scheme 15: One-pot Suzuki coupling–Buchwald–Hartwig amination.

Scheme 16: One-pot Rh/Pd-catalysed synthesis of dihydropyridobenzazepines.

Scheme 17: A retrosynthetic pathway to dibenzo[b,f]azepines via Mizoroki–Heck reaction.

Scheme 18: One-pot domino Pd-catalyzed Mizoroki–Heck–Buchwald–Hartwig synthesis of dibenzo[b,f]azepines.

Scheme 19: Dibenzo[b,f]thiapine and -oxepine synthesis via SNAr (thio)etherification, Wittig methylenation and...

Scheme 20: A retrosynthetic pathway to dibenzo[b,f]oxepines via Ullmann coupling.

Scheme 21: Ullmann-type coupling in dibenzo[b,f]oxepine synthesis.

Scheme 22: Wittig reaction and Ullmann coupling as key steps in dihydrobenz[b,f]oxepine synthesis.

Scheme 23: Pd-catalysed dibenzo[b,f]azepine synthesis via norbornene azepine intermediate 109.

Scheme 24: A simple representation of olefin metathesis resulting in transalkylidenation.

Scheme 25: Ring-closing metathesis as key step in the synthesis of dibenzo[b,f]heteropines.

Scheme 26: Alkyne–aldehyde metathesis in the synthesis of dibenzo[b,f]heteropines.

Scheme 27: Hydroarylation of 9-(2-alkynylphenyl)-9H-carbazole derivatives.

Scheme 28: Oxidative coupling of bisphonium ylide intermediate to give pacharin (13).

Scheme 29: Preparation of 10,11-dihydrodibenzo[b,f]heteropines via intramolecular Wurtz reaction.

Scheme 30: Phenol deprotonation and intramolecular etherification in the synthesis of bauhinoxepine J.

Figure 6: Functionalisation of dibenzo[b,f]azepine.

Scheme 31: Palladium-catalysed N-arylation of dibenzo[b,f]azepine.

Scheme 32: Cu- and Ni-catalysed N-arylation.

Scheme 33: N-Alkylation of dibenzo[b,f]azepine (1a) and dihydrodibenzo[b,f]azepine (2a).

Scheme 34: Preparation of methoxyiminosilbene.

Scheme 35: Synthesis of oxcarbazepine (153) from methoxy iminostilbene 151.

Scheme 36: Ring functionalisation of dihydrodibenzo[b,f]azepine.
Group 13 exchange and transborylation in catalysis
- Dominic R. Willcox and
- Stephen P. Thomas
Beilstein J. Org. Chem. 2023, 19, 325–348, doi:10.3762/bjoc.19.28
- (Scheme 16) [75]. The reaction was proposed to occur through activation of the alkyl fluoride 68 with H-B-9-BBN, followed by electrophilic substitution of the arene 69 to give a Wheland intermediate and a fluoroborohydride 70 (Scheme 16). Loss of H2 gave the arylated product 71, dihydrogen, and F-B-9-BBN

Graphical Abstract

Scheme 1: Group 13 exchange.

Scheme 2: Borane-catalysed hydroboration of alkynes and the proposed mechanism.

Scheme 3: a) Borane-catalysed hydroboration of alkenes and the proposed mechanism. b) H-B-9-BBN-catalysed dou...

Scheme 4: a) Amine-borane-catalysed C‒H borylation of heterocycles and the proposed mechanism. b) Benzoic aci...

Scheme 5: Bis(pentafluorophenyl)borane-catalysed dimerisation of allenes and the proposed mechanism.

Scheme 6: Alkoxide-promoted hydroboration of heterocycles and the proposed mechanism.

Scheme 7: Borane-catalysed reduction of indoles and the proposed mechanism.

Scheme 8: H-B-9-BBN-catalysed hydrocyanation of enones and the proposed mechanism.

Scheme 9: Borane-catalysed hydroboration of nitriles and the proposed mechanism.

Scheme 10: Myrtanylborane-catalysed asymmetric reduction of propargylic ketones and the proposed mechanism.

Scheme 11: H-B-9-BBN-catalysed C–F esterification of alkyl fluorides and the proposed mechanism.

Scheme 12: H-B-9-BBN-catalysed 1,4-hydroboration of enones and the proposed mechanism.

Scheme 13: Boric acid-promoted reduction of esters, lactones, and carbonates and the proposed mechanism.

Scheme 14: H-B-9-BBN-catalysed reductive aldol-type reaction and the proposed mechanism.

Scheme 15: H-B-9-BBN-catalysed diastereoselective allylation of ketones and the Ph-BBD-catalysed enantioselect...

Scheme 16: H-B-9-BBN-catalysed C–F arylation of benzyl fluorides and the proposed mechanism.

Scheme 17: Borane-catalysed S‒H borylation of thiols and the proposed mechanism.

Scheme 18: Borane-catalysed hydroalumination of alkenes and allenes.

Scheme 19: a) Aluminium-catalysed hydroboration of alkynes and example catalysts. b) Deprotonation mechanistic...

Scheme 20: Aluminium-catalysed hydroboration of alkenes and the proposed mechanism.

Scheme 21: Aluminium-catalysed C–H borylation of terminal alkynes and the proposed mechanism.

Scheme 22: Aluminium-catalysed dehydrocoupling of amines, alcohols, and thiols with H-B-9-BBN or HBpin and the...

Scheme 23: Aluminium-catalysed hydroboration of unsaturated compounds and the general reaction mechanism.

Scheme 24: a) Gallium-catalysed asymmetric hydroboration of ketones and the proposed mechanism. b) Gallium-cat...

Scheme 25: Gallium(I)-catalysed allylation/propargylation of acetals and aminals and the proposed mechanism.

Scheme 26: Indium(I)-catalysed allylation/propargylation of acetals, aminals, and alkyl ethers.

Scheme 27: Iron–indium cocatalysed double hydroboration of nitriles and the proposed mechanism.

Figure 1: a) The number of reports for a given group 13 exchange in catalysis. b) Average free energy barrier...
Modular synthesis of 2-furyl carbinols from 3-benzyldimethylsilylfurfural platforms relying on oxygen-assisted C–Si bond functionalization
- Sebastien Curpanen,
- Per Reichert,
- Gabriele Lupidi,
- Giovanni Poli,
- Julie Oble and
- Alejandro Perez-Luna
Beilstein J. Org. Chem. 2022, 18, 1256–1263, doi:10.3762/bjoc.18.131
- on the SiMe(OSiMe3)2 unit, which were readily converted through Pd- or Cu-catalyzed electrophilic substitution reactions into an array of furfurals decorated at C3 with carbon- or heteroatom-containing substituents (Scheme 1). Conversely, all of our subsequent efforts to achieve cross-coupling
- activation [19]. Thereby, C3-triorganosilyl-substituted furfurals could be suitable platforms to develop a two-step modular approach to 3-substituted 2-furyl carbinols, entailing nucleophilic addition to the aldehyde function and oxygen-assisted electrophilic substitution of the C–Si bond (Scheme 1). Results
- ). With the 3-silylated 2-furyl carbinol substrates at hand, we then considered C–Si bond activation strategies relying on the assistance of the oxygen atom to promote electrophilic substitution reactions with carbon electrophiles. C3–Si bond functionalization through intramolecular activation by

Graphical Abstract

Scheme 1: C3–Si bond functionalization of biomass-derived 3-silylated furfural platforms.

Scheme 2: Preparation of 3-silylated 2-furyl carbinols.

Scheme 3: C–Si bond functionalization of 2,3-disubstituted furyl carbinols by 1,4-silyl migration.

Scheme 4: Attempts of C3–Si bond functionalization promoted by intramolecular activation via alkoxide.

Scheme 5: Alkoxide-promoted cyclic siloxane formation from 2-[(3-benzyldimethylsilyl)furyl] carbinol 4c.

Scheme 6: TBAF-promoted cyclic siloxane formation from 2-[(3-benzyldimethylsilyl)furyl] carbinol 4c.

Scheme 7: Pd-catalyzed arylation of 2-[(3-benzyldimethylsilyl)furyl] carbinol 4c.

Scheme 8: Cu-catalyzed allylation and methylation of 2-[(3-benzyldimethylsilyl)furyl] carbinols. aCuI⋅PPh3 (1...
Mechanochemical halogenation of unsymmetrically substituted azobenzenes
- Dajana Barišić,
- Mario Pajić,
- Ivan Halasz,
- Darko Babić and
- Manda Ćurić
Beilstein J. Org. Chem. 2022, 18, 680–687, doi:10.3762/bjoc.18.69
- PdII catalyst and additives. These reactions in most cases produced electrophilic substitution products that were halogenated in the ortho position(s) relative to the electron-donating substituent (Scheme 1 and Table 1), as confirmed by Raman (Figures S14–S22 in Supporting Information File 1) and NMR
- dihalogenated at the ortho and meta-position(s) relative to the electron-donating substituent, which may be attributed to competition between PdII-catalyzed reactions and uncatalyzed electrophilic substitution. Our results are consistent with those reported by Sanford's group for the halogenation of various

Graphical Abstract

Figure 1: Molecular structures of the monomeric cyclopalladated intermediate and brominated product observed ...

Scheme 1: Halogenation of azobenzenes with strong electron-donating substituents.

Figure 2: a) Two-dimensional (2D) plot of the time-resolved Raman monitoring of NG of L2 (0.50 mmol) with NBS...

Figure 3: Experimental X-ray molecular structure of succinimide product L4-III.

Scheme 2: PdII-catalyzed halogenation of azobenzene and its para-halogenated derivatives.

Figure 4: Experimental X-ray molecular structure of the intermediate I6-I.

Figure 5: a) In situ observation of I6-I during the time-resolved Raman monitoring of LAG of L6 (0.50 mmol) w...
Menadione: a platform and a target to valuable compounds synthesis
- Acácio S. de Souza,
- Ruan Carlos B. Ribeiro,
- Dora C. S. Costa,
- Fernanda P. Pauli,
- David R. Pinho,
- Matheus G. de Moraes,
- Fernando de C. da Silva,
- Luana da S. M. Forezi and
- Vitor F. Ferreira
Beilstein J. Org. Chem. 2022, 18, 381–419, doi:10.3762/bjoc.18.43
- aldehydes and ketones [128]. In this context, Fry and co-workers explored the electrophilic substitution reaction to synthesize 2-methyl-3-bromonaphthalene-1,4-dione (82), an important intermediate used for the synthesis of naphthoquinones functionalized with organochalcogens [127]. Compound 82 was obtained

Graphical Abstract

Figure 1: Natural bioactive naphthoquinones.

Figure 2: Chemical structures of vitamins K.

Figure 3: Redox cycle of menadione.

Scheme 1: Selected approaches for menadione synthesis using silver(I) as a catalyst.

Scheme 2: Methylation approaches for the preparation of menadione from 1,4-naphthoquinone using tert-butyl hy...

Scheme 3: Methylation approach of 1,4-naphthoquinone using i) rhodium complexes/methylboronic acid and ii) bi...

Scheme 4: Synthesis of menadione (10) from itaconic acid.

Scheme 5: Menadione synthesis via Diels–Alder reaction.

Scheme 6: Synthesis of menadione (10) using p-cresol as a synthetic precursor.

Scheme 7: Synthesis of menadione (10) via demethoxycarbonylating annulation of methyl methacrylate.

Scheme 8: Furan 34 used as a diene in a Diels–Alder reaction for the synthesis of menadione (10).

Scheme 9: o-Toluidine as a dienophile in a Diels–Alder reaction for the synthesis of menadione (10).

Scheme 10: Representation of electrochemical synthesis of menadione.

Figure 4: Reaction sites and reaction types of menadione as substrate.

Scheme 11: DBU-catalyzed epoxidation of menadione (10).

Scheme 12: Phase-transfer catalysis for the epoxidation of menadione.

Scheme 13: Menadione epoxidation using a hydroperoxide derived from (+)-norcamphor.

Scheme 14: Enantioselective Diels–Alder reaction for the synthesis of asymmetric quinone 50 catalyzed by a chi...

Scheme 15: Optimized reaction conditions for the synthesis of anthra[9,1-bc]pyranone.

Scheme 16: Synthesis of anthra[9,1-bc]furanone, anthra[9,1-bc]pyridine, and anthra[9,1-bc]pyrrole derivatives.

Scheme 17: Synthesis of derivatives employing protected trienes.

Scheme 18: Synthesis of cyclobutene derivatives of menadione.

Scheme 19: Menadione reduction reactions using sodium hydrosulfite.

Scheme 20: Green methodology for menadiol synthesis and pegylation.

Scheme 21: Menadione reduction by 5,6-O-isopropylidene-ʟ-ascorbic acid under UV light irradiation.

Scheme 22: Selected approaches of menadione hydroacetylation to diacetylated menadiol.

Scheme 23: Thiele–Winter reaction catalyzed by Bi(OTf)3.

Scheme 24: Carbonyl condensation of menadione using resorcinol and a hydrazone derivative.

Scheme 25: Condensation reaction of menadione with thiosemicarbazide.

Scheme 26: Condensation reaction of menadione with acylhydrazides.

Scheme 27: Menadione derivatives functionalized with organochalcogens.

Scheme 28: Synthesis of selenium-menadione conjugates derived from chloromethylated menadione 84.

Scheme 29: Menadione alkylation by the Kochi–Anderson method.

Scheme 30: Menadione alkylation by diacids.

Scheme 31: Menadione alkylation by heterocycles-substituted carboxylic acids.

Scheme 32: Menadione alkylation by bromoalkyl-substituted carboxylic acids.

Scheme 33: Menadione alkylation by complex carboxylic acids.

Scheme 34: Kochi–Anderson method variations for the menadione alkylation via oxidative decarboxylation of carb...

Scheme 35: Copper-catalyzed menadione alkylation via free radicals.

Scheme 36: Nickel-catalyzed menadione cyanoalkylation.

Scheme 37: Iron-catalyzed alkylation of menadione.

Scheme 38: Selected approaches to menadione alkylation.

Scheme 39: Menadione acylation by photo-Friedel–Crafts acylation reported by Waske and co-workers.

Scheme 40: Menadione acylation by Westwood procedure.

Scheme 41: Synthesis of 3-benzoylmenadione via metal-free TBAI/TBHP system.

Scheme 42: Michael-type addition of amines to menadione reported by Kallmayer.

Scheme 43: Synthesis of amino-menadione derivatives using polyalkylamines.

Scheme 44: Selected examples for the synthesis of different amino-substituted menadione derivatives.

Scheme 45: Selected examples of Michael-type addition of complex amines to menadione (10).

Scheme 46: Addition of different natural α-amino acids to menadione.

Scheme 47: Michael-type addition of amines to menadione using silica-supported perchloric acid.

Scheme 48: Indolylnaphthoquinone or indolylnaphthalene-1,4-diol synthesis reported by Yadav et al.

Scheme 49: Indolylnaphthoquinone synthesis reported by Tanoue and co-workers.

Scheme 50: Indolylnaphthoquinone synthesis from menadione by Escobeto-González and co-workers.

Scheme 51: Synthesis of menadione analogues functionalized with thiols.

Scheme 52: Synthesis of menadione-derived symmetrical derivatives through reaction with dithiols.

Scheme 53: Mercaptoalkyl acids as nucleophiles in Michael-type addition reaction to menadione.

Scheme 54: Reactions of menadione (10) with cysteine derivatives for the synthesis of quinoproteins.

Scheme 55: Synthesis of menadione-glutathione conjugate 152 by Michael-type addition.
AlBr3-Promoted stereoselective anti-hydroarylation of the acetylene bond in 3-arylpropynenitriles by electron-rich arenes: synthesis of 3,3-diarylpropenenitriles
- Yelizaveta Gorbunova,
- Dmitry S. Ryabukhin and
- Aleksander V. Vasilyev
Beilstein J. Org. Chem. 2021, 17, 2663–2667, doi:10.3762/bjoc.17.180
- hydroarylation of nitriles 1. Reactions of nitriles 1a–c with o-xylene led to the formation of regioisomers derived from the electrophilic substitution at different positions of this arene. Thus, nitrile 1a gave two types of regioisomers 2n and 2o. After reactions of nitriles 1b,c with o-xylene, compounds 2l and

Graphical Abstract

Scheme 1: AlBr3-promoted hydroarylation of the acetylene bond of 3-arylpropynenitriles 1a–c by arenes with th...

Scheme 2: Plausible mechanism for reaction of acetylene nitriles 1 with arenes leading to nitriles 2.

Scheme 3: Cyclization of nitriles 2c,g into indanones 3a,b in TfOH.

Scheme 4: Hydrophenylation of nitriles 1a,b by benzene in TfOH leading to nitriles 2a,b.
On the application of 3d metals for C–H activation toward bioactive compounds: The key step for the synthesis of silver bullets
- Renato L. Carvalho,
- Amanda S. de Miranda,
- Mateus P. Nunes,
- Roberto S. Gomes,
- Guilherme A. M. Jardim and
- Eufrânio N. da Silva Júnior
Beilstein J. Org. Chem. 2021, 17, 1849–1938, doi:10.3762/bjoc.17.126

- heteroaromatic electrophilic substitution and a non-radical pathway. An aminomethylation of the heteroaromatic ring with N-methylmorpholine-N-oxide catalyzed by VO(aca)2 reported by Mitchell and co-workers [109], however, was found to undergo with a regioselective outcome incompatible to an electrophilic

Graphical Abstract

Scheme 1: Schematic overview of transition metals studied in C–H activation processes.

Scheme 2: (A) Known biological activities related to benzimidazole-based compounds; (B and C) an example of a...

Scheme 3: (A) Known biological activities related to quinoline-based compounds; (B and C) an example of a sca...

Scheme 4: (A) Known biological activities related to sulfur-containing compounds; (B and C) an example of a s...

Scheme 5: (A) Known biological activities related to aminoindane derivatives; (B and C) an example of a scand...

Scheme 6: (A) Known biological activities related to norbornane derivatives; (B and C) an example of a scandi...

Scheme 7: (A) Known biological activities related to aniline derivatives; (B and C) an example of a titanium-...

Scheme 8: (A) Known biological activities related to cyclohexylamine derivatives; (B) an example of an intram...

Scheme 9: (A) Known biologically active benzophenone derivatives; (B and C) photocatalytic oxidation of benzy...

Scheme 10: (A) Known bioactive fluorine-containing compounds; (B and C) vanadium-mediated C(sp3)–H fluorinatio...

Scheme 11: (A) Known biologically active Lythraceae alkaloids; (B) synthesis of (±)-decinine (30).

Scheme 12: (A) Synthesis of (R)- and (S)-boehmeriasin (31); (B) synthesis of phenanthroindolizidines by vanadi...

Scheme 13: (A) Known bioactive BINOL derivatives; (B and C) vanadium-mediated oxidative coupling of 2-naphthol...

Scheme 14: (A) Known antiplasmodial imidazopyridazines; (B) practical synthesis of 41.

Scheme 15: (A) Gold-catalyzed drug-release mechanism using 2-alkynylbenzamides; (B and C) chromium-mediated al...

Scheme 16: (A) Examples of anti-inflammatory benzaldehyde derivatives; (B and C) chromium-mediated difunctiona...

Scheme 17: (A and B) Manganese-catalyzed chemoselective intramolecular C(sp3)–H amination; (C) late-stage modi...

Scheme 18: (A and B) Manganese-catalyzed C(sp3)–H amination; (C) late-stage modification of a leelamine deriva...

Scheme 19: (A) Known bioactive compounds containing substituted N-heterocycles; (B and C) manganese-catalyzed ...

Scheme 20: (A) Known indoles that present GPR40 full agonist activity; (B and C) manganese-catalyzed C–H alkyl...

Scheme 21: (A) Examples of known biaryl-containing drugs; (B and C) manganese-catalyzed C–H arylation through ...

Scheme 22: (A) Known zidovudine derivatives with potent anti-HIV properties; (B and C) manganese-catalyzed C–H...

Scheme 23: (A and B) Manganese-catalyzed C–H organic photo-electrosynthesis; (C) late-stage modification.

Scheme 24: (A) Example of a known antibacterial silylated dendrimer; (B and C) manganese-catalyzed C–H silylat...

Scheme 25: (A and B) Fe-based small molecule catalyst applied for selective aliphatic C–H oxidations; (C) late...

Scheme 26: (A) Examples of naturally occurring gracilioethers; (B) the first total synthesis of gracilioether ...

Scheme 27: (A and B) Selective aliphatic C–H oxidation of amino acids; (C) late-stage modification of proline-...

Scheme 28: (A) Examples of Illicium sesquiterpenes; (B) first chemical synthesis of (+)-pseudoanisatin (80) in...

Scheme 29: (A and B) Fe-catalyzed deuteration; (C) late-stage modification of pharmaceuticals.

Scheme 30: (A and B) Biomimetic Fe-catalyzed aerobic oxidation of methylarenes to benzaldehydes (PMHS, polymet...

Scheme 31: (A) Known tetrahydroquinolines with potential biological activities; (B and C) redox-selective Fe c...

Scheme 32: (A) Known drugs containing a benzofuran unit; (B and C) Fe/Cu-catalyzed tandem O-arylation to acces...

Scheme 33: (A) Known azaindolines that act as M4 muscarinic acetylcholine receptor agonists; (B and C) intramo...

Scheme 34: (A) Known indolinones with anticholinesterase activity; (B and C) oxidative C(sp3)–H cross coupling...

Scheme 35: (A and B) Cobalt-catalyzed C–H alkenylation of C-3-peptide-containing indoles; (C) derivatization b...

Scheme 36: (A) Cobalt-Cp*-catalyzed C–H methylation of known drugs; (B and C) scope of the o-methylated deriva...

Scheme 37: (A) Known lasalocid A analogues; (B and C) three-component cobalt-catalyzed C–H bond addition; (D) ...

Scheme 38: (A and B) Cobalt-catalyzed C(sp2)–H amidation of thiostrepton.

Scheme 39: (A) Known 4H-benzo[d][1,3]oxazin-4-one derivatives with hypolipidemic activity; (B and C) cobalt-ca...

Scheme 40: (A and B) Cobalt-catalyzed C–H arylation of pyrrole derivatives; (C) application for the synthesis ...

Scheme 41: (A) Known 2-phenoxypyridine derivatives with potent herbicidal activity; (B and C) cobalt-catalyzed...

Scheme 42: (A) Natural cinnamic acid derivatives; (B and C) cobalt-catalyzed C–H carboxylation of terminal alk...

Scheme 43: (A and B) Cobalt-catalyzed C–H borylation; (C) application to the synthesis of flurbiprofen.

Scheme 44: (A) Benzothiazoles known to present anticonvulsant activities; (B and C) cobalt/ruthenium-catalyzed...

Scheme 45: (A and B) Cobalt-catalyzed oxygenation of methylene groups towards ketone synthesis; (C) synthesis ...

Scheme 46: (A) Known anticancer tetralone derivatives; (B and C) cobalt-catalyzed C–H difluoroalkylation of ar...

Scheme 47: (A and B) Cobalt-catalyzed C–H thiolation; (C) application in the synthesis of quetiapine (153).

Scheme 48: (A) Known benzoxazole derivatives with anticancer, antifungal, and antibacterial activities; (B and...

Scheme 49: (A and B) Cobalt-catalyzed C–H carbonylation of naphthylamides; (C) BET inhibitors 158 and 159 tota...

Scheme 50: (A) Known bioactive pyrrolo[1,2-a]quinoxalin-4(5H)-one derivatives; (B and C) cobalt-catalyzed C–H ...

Scheme 51: (A) Known antibacterial cyclic sulfonamides; (B and C) cobalt-catalyzed C–H amination of propargyli...

Scheme 52: (A and B) Cobalt-catalyzed intramolecular 1,5-C(sp3)–H amination; (C) late-stage functionalization ...

Scheme 53: (A and B) Cobalt-catalyzed C–H/C–H cross-coupling between benzamides and oximes; (C) late-state syn...

Scheme 54: (A) Known anticancer natural isoquinoline derivatives; (B and C) cobalt-catalyzed C(sp2)–H annulati...

Scheme 55: (A) Enantioselective intramolecular nickel-catalyzed C–H activation; (B) bioactive obtained motifs;...

Scheme 56: (A and B) Nickel-catalyzed α-C(sp3)–H arylation of ketones; (C) application of the method using kno...

Scheme 57: (A and B) Nickel-catalyzed C(sp3)–H acylation of pyrrolidine derivatives; (C) exploring the use of ...

Scheme 58: (A) Nickel-catalyzed C(sp3)–H arylation of dioxolane; (B) library of products obtained from biologi...

Scheme 59: (A) Intramolecular enantioselective nickel-catalyzed C–H cycloalkylation; (B) product examples, inc...

Scheme 60: (A and B) Nickel-catalyzed C–H deoxy-arylation of azole derivatives; (C) late-stage functionalizati...

Scheme 61: (A and B) Nickel-catalyzed decarbonylative C–H arylation of azole derivatives; (C) application of t...

Scheme 62: (A and B) Another important example of nickel-catalyzed C–H arylation of azole derivatives; (C) app...

Scheme 63: (A and B) Another notable example of a nickel-catalyzed C–H arylation of azole derivatives; (C) lat...

Scheme 64: (A and B) Nickel-based metalorganic framework (MOF-74-Ni)-catalyzed C–H arylation of azole derivati...

Scheme 65: (A) Known commercially available benzothiophene-based drugs; (B and C) nickel-catalyzed C–H arylati...

Scheme 66: (A) Known natural tetrahydrofuran-containing substances; (B and C) nickel-catalyzed photoredox C(sp3...

Scheme 67: (A and B) Another notable example of a nickel-catalyzed photoredox C(sp3)–H alkylation/arylation; (...

Scheme 68: (A) Electrochemical/nickel-catalyzed C–H alkoxylation; (B) achieved scope, including three using na...

Scheme 69: (A) Enantioselective photoredox/nickel catalyzed C(sp3)–H arylation; (B) achieved scope, including ...

Scheme 70: (A) Known commercially available trifluoromethylated drugs; (B and C) nickel-catalyzed C–H trifluor...

Scheme 71: (A and B) Stereoselective nickel-catalyzed C–H difluoroalkylation; (C) late-stage functionalization...

Scheme 72: (A) Cu-mediated ortho-amination of oxalamides; (B) achieved scope, including derivatives obtained f...

Scheme 73: (A) Electro-oxidative copper-mediated amination of 8-aminoquinoline-derived amides; (B) achieved sc...

Scheme 74: (A and B) Cu(I)-mediated C–H amination with oximes; (C) derivatization using telmisartan (241) as s...

Scheme 75: (A and B) Cu-mediated amination of aryl amides using ammonia; (C) late-stage modification of proben...

Scheme 76: (A and B) Synthesis of purine nucleoside analogues using copper-mediated C(sp2)–H activation.

Scheme 77: (A) Copper-mediated annulation of acrylamide; (B) achieved scope, including the synthesis of the co...

Scheme 78: (A) Known bioactive compounds containing a naphthyl aryl ether motif; (B and C) copper-mediated eth...

Scheme 79: (A and B) Cu-mediated alkylation of N-oxide-heteroarenes; (C) late-stage modification.

Scheme 80: (A) Cu-mediated cross-dehydrogenative coupling of polyfluoroarenes and alkanes; (B) scope from know...

Scheme 81: (A) Known anticancer acrylonitrile compounds; (B and C) Copper-mediated cyanation of unactivated al...

Scheme 82: (A) Cu-mediated radiofluorination of 8-aminoquinoline-derived aryl amides; (B) achieved scope, incl...

Scheme 83: (A) Examples of natural β-carbolines; (B and C) an example of a zinc-catalyzed C–H functionalizatio...

Scheme 84: (A) Examples of anticancer α-aminophosphonic acid derivatives; (B and C) an example of a zinc-catal...
Iodine-catalyzed electrophilic substitution of indoles: Synthesis of (un)symmetrical diindolylmethanes with a quaternary carbon center
- Thanigaimalai Pillaiyar,
- Masoud Sedaghati,
- Andhika B. Mahardhika,
- Lukas L. Wendt and
- Christa E. Müller
Beilstein J. Org. Chem. 2021, 17, 1464–1475, doi:10.3762/bjoc.17.102
- derivatives have recently received increasing attention from synthetic organic chemists, biologists, and pharmacologists. In general, DIMs can be synthesized via electrophilic substitution of indoles by aldehydes or ketones in the presence of conventional Lewis or Brønsted acids as catalysts [19]. This

Graphical Abstract

Figure 1: Diindolylmethanes and reported biological activities.

Figure 2: Synthetic strategies toward trifluoromethylated unsymmetrical quaternary DIMs.

Figure 3: Reactions performed to study the scope of the method.

Figure 4: Gram-scale synthesis of unsymmetrical DIMs 3a and 3ad.

Figure 5: Plausible reaction mechanism for the synthesis of fluoromethylated unsymmetrical DIMs, shown for co...
Synthetic accesses to biguanide compounds
- Oleksandr Grytsai,
- Cyril Ronco and
- Rachid Benhida
Beilstein J. Org. Chem. 2021, 17, 1001–1040, doi:10.3762/bjoc.17.82

- , the authors observed an intramolecular aromatic electrophilic substitution under high-temperature conditions. This unprecedented reactivity was used in the synthesis of 1,3-diaminobenzoquinazoline derivatives (Scheme 26). After prolonged reflux in octanol, the quinazolines were isolated as free bases

Graphical Abstract

Figure 1: Tautomeric forms of biguanide.

Figure 2: Illustrations of neutral, monoprotonated, and diprotonated structures biguanide.

Figure 3: The main approaches for the synthesis of biguanides. The core structure is obtained via the additio...

Scheme 1: The three main preparations of biguanides from cyanoguanidine.

Scheme 2: Synthesis of butylbiguanide using CuCl2 [16].

Scheme 3: Synthesis of biguanides by the direct fusion of cyanoguanidine and amine hydrochlorides [17,18].

Scheme 4: Synthesis of ethylbiguanide and phenylbiguanide as reported by Smolka and Friedreich [14].

Scheme 5: Synthesis of arylbiguanides through the reaction of cyanoguanidine with anilines in water [19].

Scheme 6: Synthesis of aryl- and alkylbiguanides by adaptations of Cohn’s procedure [20,21].

Scheme 7: Microwave-assisted synthesis of N1-aryl and -dialkylbiguanides [22,23].

Scheme 8: Synthesis of aryl- and alkylbiguanides by trimethylsilyl activation [24,26].

Scheme 9: Synthesis of phenformin analogs by TMSOTf activation [27].

Scheme 10: Synthesis of N1-(1,2,4-triazolyl)biguanides [28].

Scheme 11: Synthesis of 2-guanidinobenzazoles by addition of ortho-substituted anilines to cyanoguanidine [30,32] and...

Scheme 12: Synthesis of 2,4-diaminoquinazolines by the addition of 2-cyanoaniline to cyanoguanidine and from 3...

Scheme 13: Reactions of anthranilic acid and 2-mercaptobenzoic acid with cyanoguanidine [24,36,37].

Scheme 14: Synthesis of disubstituted biguanides with Cu(II) salts [38].

Scheme 15: Synthesis of an N1,N2,N5-trisubstituted biguanide by fusion of an amine hydrochloride and 2-cyano-1...

Scheme 16: Synthesis of N1,N5-disubstituted biguanides by the addition of anilines to cyanoguanidine derivativ...

Scheme 17: Microwave-assisted additions of piperazine and aniline hydrochloride to substituted cyanoguanidines ...

Scheme 18: Synthesis of N1,N5-alkyl-substituted biguanides by TMSOTf activation [27].

Scheme 19: Additions of oxoamines hydrochlorides to dimethylcyanoguanidine [49].

Scheme 20: Unexpected cyclization of pyridylcyanoguanidines under acidic conditions [50].

Scheme 21: Example of industrial synthesis of chlorhexidine [51].

Scheme 22: Synthesis of symmetrical N1,N5-diarylbiguanides from sodium dicyanamide [52,53].

Scheme 23: Synthesis of symmetrical N1,N5-dialkylbiguanides from sodium dicyanamide [54-56].

Scheme 24: Stepwise synthesis of unsymmetrical N1,N5-trisubstituted biguanides from sodium dicyanamide [57].

Scheme 25: Examples for the synthesis of unsymmetrical biguanides [58].

Scheme 26: Examples for the synthesis of an 1,3-diaminobenzoquinazoline derivative by the SEAr cyclization of ...

Scheme 27: Major isomers formed by the SEAr cyclization of symmetric biguanides derived from 2- and 3-aminophe...

Scheme 28: Lewis acid-catalyzed synthesis of 8H-pyrrolo[3,2-g]quinazoline-2,4-diamine [63].

Scheme 29: Synthesis of [1,2,4]oxadiazoles by the addition of hydroxylamine to dicyanamide [49,64].

Scheme 30: Principle of “bisamidine transfer” and analogy between the reactions with N-amidinopyrazole and N-a...

Scheme 31: Representative syntheses of N-amidino-amidinopyrazole hydrochloride [68,69].

Scheme 32: First examples of biguanide syntheses using N-amidino-amidinopyrazole [66].

Scheme 33: Example of “biguanidylation” of a hydrazide substrate [70].

Scheme 34: Example for the synthesis of biguanides using S-methylguanylisothiouronium iodide as “bisamidine tr...

Scheme 35: Synthesis of N-substituted N1-cyano-S-methylisothiourea precursors.

Scheme 36: Addition routes on N1-cyano-S-methylisothioureas.

Scheme 37: Synthesis of an hydroxybiguanidine from N1-cyano-S-methylisothiourea [77].

Scheme 38: Synthesis of an N1,N2,N3,N4,N5-pentaarylbiguanide from the corresponding triarylguanidine and carbo...

Scheme 39: Reactions of N,N,N’,N’-tetramethylguanidine (TMG) with carbodiimides to synthesize hexasubstituted ...

Scheme 40: Microwave-assisted addition of N,N,N’,N’-tetramethylguanidine to carbodiimides [80].

Scheme 41: Synthesis of N1-aryl heptasubstituted biguanides via a one-pot biguanide formation–copper-catalyzed ...

Scheme 42: Formation of 1,2-dihydro-1,3,5-triazine derivatives by the reaction of guanidine with excess carbod...

Scheme 43: Plausible mechanism for the spontaneous cyclization of triguanides [82].

Scheme 44: a) Formation of mono- and disubstituted (iso)melamine derivatives by the reaction of biguanides and...

Scheme 45: Reactions of 2-aminopyrimidine with carbodiimides to synthesize 2-guanidinopyrimidines as “biguanid...

Scheme 46: Non-catalyzed alternatives for the addition of 2-aminopyrimidine derivatives to carbodiimides. A) h...

Scheme 47: Addition of guanidinomagnesium halides to substituted cyanamides [90].

Scheme 48: Microwave-assisted synthesis of [11C]metformin by the reaction of 11C-labelled dimethylcyanamide an...

Scheme 49: Formation of 4-amino-6-dimethylamino[1,3,5]triazin-2-ol through the reaction of Boc-guanidine and d...

Scheme 50: Formation of 1,3,5-triazine derivatives via the addition of guanidines to substituted cyanamides [92].

Scheme 51: Synthesis of biguanide by the reaction of O-alkylisourea and guanidine [93].

Scheme 52: Aromatic nucleophilic substitution of guanidine on 2-O-ethyl-1,3,5-triazine [95].

Scheme 53: Synthesis of N1,N2-disubstituted biguanides by the reaction of guanidine and thioureas in the prese...

Scheme 54: Cyclization reactions involving condensations of guanidine(-like) structures with thioureas [97,98].

Scheme 55: Condensations of guanidine-like structures with thioureas [99,100].

Scheme 56: Condensations of guanidines with S-methylisothioureas [101,102].

Scheme 57: Addition of 2-amino-1,3-diazaaromatics to S-alkylisothioureas [103,104].

Scheme 58: Addition of guanidines to 2-(methylsulfonyl)pyrimidines [105].

Scheme 59: An example of a cyclodesulfurization reaction to a fused 3,5-diamino-1,2,4-triazole [106].

Scheme 60: Ring-opening reactions of 1,3-diaryl-2,4-bis(arylimino)-1,3-diazetidines [107].

Scheme 61: Formation of 3,5-diamino-1,2,4-triazole derivatives via addition of hydrazines to 1,3-diazetidine-2...

Scheme 62: Formation of a biguanide via the addition of aniline to 1,2,4-thiadiazol-3,5-diamines, ring opening...

Figure 4: Substitution pattern of biguanides accessible by synthetic pathways a–h.
Application of the Meerwein reaction of 1,4-benzoquinone to a metal-free synthesis of benzofuropyridine analogues
- Rashmi Singh,
- Tomas Horsten,
- Rashmi Prakash,
- Swapan Dey and
- Wim Dehaen
Beilstein J. Org. Chem. 2021, 17, 977–982, doi:10.3762/bjoc.17.79
- . Electrophilic substitution and further condensations give polycyclic systems, including oxazolo- and chromeno-fused analogues. Keywords: benzofuropyridines; benzoquinones; dibenzofurans; Meerwein reaction; metal-free synthesis; Introduction Dibenzofurans are important oxygen-containing heterocycles present in

Graphical Abstract

Figure 1: Biologically relevant 2-oxydibenzofuran-containing structures 1–6.

Figure 2: Representative bioactive structures containing benzofuro-fused pyridine analogues 7–9.

Scheme 1: Strategy for metal-free access to benzofuropyridine 13.

Scheme 2: Electrophilic aromatic substitution of 6-hydroxybenzofuro[2,3-b]pyridine (13).

Scheme 3: Synthesis of isomeric oxazole-fused derivatives.

Scheme 4: Fused derivatives from 16.
All-carbon [3 + 2] cycloaddition in natural product synthesis
- Zhuo Wang and
- Junyang Liu
Beilstein J. Org. Chem. 2020, 16, 3015–3031, doi:10.3762/bjoc.16.251

- tetrachloride produces an alkyoxy allylic carbocation (not shown). This carbocation is subjected to a regiospecific electrophilic substitution of allene 168 to generate a vinyl cation 172, which is stabilized by an adjacent carbon–silicon bond. The 1,2-shift of the silyl group in 172 produces an isomeric vinyl

Graphical Abstract

Figure 1: Highly-substituted five-membered carbocycle in biologically significant natural products.

Figure 2: Natural product synthesis featuring the all-carbon [3 + 2] cycloaddition. (Quaternary carbon center...

Scheme 1: Representative natural product syntheses that feature the all-carbon [3 + 2] cyclization as the key...

Scheme 2: (A) An intramolecular trimethylenemethane diyl [3 + 2] cycloaddition with allenyl diazo compound 38...

Scheme 3: (A) Palladium-catalyzed intermolecular carboxylative TMM cycloaddition [36]. (B) The proposed mechanism....

Scheme 4: Natural product syntheses that make use of palladium-catalyzed intermolecular [3 + 2] cycloaddition...

Scheme 5: (A) Phosphine-catalyzed [3 + 2] cycloaddition [17]. (B) The proposed mechanism.

Scheme 6: Lu’s [3 + 2] cycloaddition in natural product synthesis. (A) Synthesis of longeracinphyllin A (10) [41]...

Scheme 7: (A) Phosphine-catalyzed [3 + 2] annulation of unsymmetric isoindigo 100 with allene in the preparat...

Scheme 8: (A) Rhodium-catalyzed intracmolecular [3 + 2] cycloaddition [49]. (B) The proposed catalytic cycle of t...

Scheme 9: Total synthesis of natural products reported by Yang and co-workers applying rhodium-catalyzed intr...

Scheme 10: (A) Platinum(II)-catalyzed intermolecular [3 + 2] cycloaddition of propargyl ether 139 and n-butyl ...

Scheme 11: (A) Platinum-catalyzed intramolecular [3 + 2] cycloaddition of propargylic ketal derivative 142 to ...

Scheme 12: (A) Synthesis of phyllocladanol (21) features a Lewis acid-catalyzed formal intramolecular [3 + 2] ...

Scheme 13: The recent advances of [3 + 2] annulation in natural product synthesis. (A) The preparation of melo...
Three-component reactions of aromatic amines, 1,3-dicarbonyl compounds, and α-bromoacetaldehyde acetal to access N-(hetero)aryl-4,5-unsubstituted pyrroles
- Wenbo Huang,
- Kaimei Wang,
- Ping Liu,
- Minghao Li,
- Shaoyong Ke and
- Yanlong Gu
Beilstein J. Org. Chem. 2020, 16, 2920–2928, doi:10.3762/bjoc.16.241
- enamine intermediate I to generate another intermediate III. Subsequently, III underwent an intramolecular electrophilic substitution to form the intermediate IV. Finally, IV underwent an elimination of HBr and a spontaneous aromatization to afford the pyrrole product 4a [48][49]. Apart from the pyrrole

Graphical Abstract

Figure 1: Representative biologically active N-(hetero)aryl-4,5-unsubstituted pyrrole scaffolds.

Scheme 1: Typical routes to N-(heteroaryl)-4,5-unsubstituted pyrroles.

Scheme 2: Substrate scope of the pyrrole synthesis.

Scheme 3: Synthesis of N-heterocyclic pyrroles.

Scheme 4: Direct synthesis of pyrrole-3-carboxamide derivatives.

Scheme 5: Plausible mechanism of the three-component reaction.

Scheme 6: Synthesis of polysubstituted pyrazolo[3,4-b]pyridine derivatives.
Rearrangement of o-(pivaloylaminomethyl)benzaldehydes: an experimental and computational study
- Csilla Hargitai,
- Györgyi Koványi-Lax,
- Tamás Nagy,
- Péter Ábrányi-Balogh,
- András Dancsó,
- Gábor Tóth,
- Judit Halász,
- Angéla Pandur,
- Gyula Simig and
- Balázs Volk
Beilstein J. Org. Chem. 2020, 16, 1636–1648, doi:10.3762/bjoc.16.136
- via their ring tautomers 18a,b. The mechanism proposed for the formation of the intermediates 15a,b and 17a,b is supported by the fact that electrophilic substitution reactions of isoindole are well known in the literature [3][13][14][15][16][17][18] and these are analogous to that suggested for the

Graphical Abstract

Scheme 1: Rearrangement of methylenedioxy-substituted aminoaldehyde 1a to regioisomer 2a and formation of the...

Scheme 2: Synthesis of 1-arylisoindoles 6 and formation of dimers 5.

Scheme 3: Rearrangement of aminoaldehydes 1 to regioisomers 2 and formation of dimer-like products 3 and 8.

Figure 1: X-ray structures of compounds 3b (left) and 8b (right).

Scheme 4: Proposed mechanism of the isomerization of aldehydes 1 via isoindoles 4.

Scheme 5: Proposed mechanism of the formation of dimer-like products 3a,b.

Scheme 6: Proposed mechanism for the formation of dimer-like products 8a,b.

Scheme 7: Dimerization of indole under acidic conditions.

Figure 2: Gibbs free energy diagram for the 1→2 rearrangement.

Scheme 8: Reaction of o-(pivaloylaminomethyl)benzaldehyde (1e) to give dimer-like products 23a and 23b.

Figure 3: X-ray structures of compounds 23a (left) and 23b (right).

Figure 4: Structures of the minimal energy conformer of stereoisomer 23a and those of two minimal energy conf...
In silico rationalisation of selectivity and reactivity in Pd-catalysed C–H activation reactions
- Liwei Cao,
- Mikhail Kabeshov,
- Steven V. Ley and
- Alexei A. Lapkin
Beilstein J. Org. Chem. 2020, 16, 1465–1475, doi:10.3762/bjoc.16.122
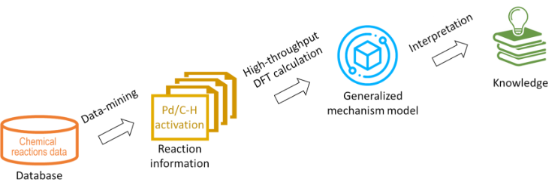
- , explaining the experimental observations for C–H activation reactions, depending on the nature of a ligand (Ln) and transition metal (M) in the catalytically active species (LnM). These mechanisms include four elementary steps: oxidative addition, σ-bond metathesis, electrophilic substitution and 1,2
- a relatively unstable intermediate (Figure 1) [26], the Hammond postulate can be applied to the electrophilic substitution reactions. The Hammond postulate states that a transition state will be structurally and energetically similar to the species (reactant, intermediate or product) nearest to it

Graphical Abstract

Figure 1: An approximate energy map for the electrophilic aromatic substitution mechanism.

Scheme 1: Schematic representation of the two mechanisms of Pd-catalysed C–H activation reaction considered i...
Synthesis and anticancer activity of bis(2-arylimidazo[1,2-a]pyridin-3-yl) selenides and diselenides: the copper-catalyzed tandem C–H selenation of 2-arylimidazo[1,2-a]pyridine with selenium
- Mio Matsumura,
- Tsutomu Takahashi,
- Hikari Yamauchi,
- Shunsuke Sakuma,
- Yukako Hayashi,
- Tadashi Hyodo,
- Tohru Obata,
- Kentaro Yamaguchi,
- Yasuyuki Fujiwara and
- Shuji Yasuike
Beilstein J. Org. Chem. 2020, 16, 1075–1083, doi:10.3762/bjoc.16.94

- experiments, a plausible double selenation mechanism is shown in Scheme 2 and Scheme 3. The first step of the reaction involves the generation of the intermediate B via A by the Cu-mediated electrophilic substitution of 1 with selenium. The oxidative homocoupling of the intermediate B then proceeds to give

Graphical Abstract

Figure 1: Biologically active selenides and diselenides having heteroaryl groups.

Figure 2: Ortep drawings of 2a (a and b) and 3a (c and d, thermal elipsoids indicate 50% probability).

Figure 3: The synthesis of bis(2-arylimidazopyridin-3-yl) diselenides. Reaction conditions: 1 (2 mmol), Se (2...

Figure 4: The synthesis of bis(2-arylimidazopyridin-3-yl) selenides. Reaction conditions: 1 (2 mmol), Se (1 m...

Scheme 1: Control reactions.

Scheme 2: Proposed mechanism (1).

Scheme 3: Proposed mechanism (2).

Figure 5: The cytotoxic effect of the bis(2-arylimidazo[1,2-a]pyridin-3-yl) diselenides 2 and selenides 3 on ...

Figure 6: The cytotoxic effect of the bis[2-(4-methoxyphenyl)imidazo[1,2-a]pyridin-3-yl] diselenide 2f on can...

Figure 7: Cytotoxic effect of the bis[2-(4-methoxyphenyl)imidazo[1,2-a]pyridin-3-yl] diselenide 2f on a cance...
Acid-catalyzed rearrangements in arenes: interconversions in the quaterphenyl series
- Sarah L. Skraba-Joiner,
- Carter J. Holt and
- Richard P. Johnson
Beilstein J. Org. Chem. 2019, 15, 2655–2663, doi:10.3762/bjoc.15.258
- believed to occur through the intermediacy of ipso arenium ions 4–6 which connect through 1,2-phenyl shifts. The term "ipso" was first proposed by Perrin and Skinner to explain unusual results in electrophilic substitution reactions; this refers to protonation at the site of a substituent [24]. Ipso

Graphical Abstract

Scheme 1: Acid-catalyzed rearrangements of arenes.

Scheme 2: Rearrangement of quaterphenyl isomers by phenyl shifts.

Scheme 3: Synthesis of quaterphenyl isomers.

Scheme 4: Rearrangement of quaterphenyl isomers via (a) 1,2-phenyl shift and (b) 1,2-biphenyl shift.

Figure 1: Pathways for terminal 1,2-phenyl shifts in quaterphenyl isomers calculated with IEFPCM(DCE)/B3LYP/6...

Figure 2: Pathways for 1,2-biphenyl shifts in quaterphenyl isomers calculated with IEFPCM(DCE)/B3LYP/6-31+G(d...
A review of the total syntheses of triptolide
- Xiang Zhang,
- Zaozao Xiao and
- Hongtao Xu
Beilstein J. Org. Chem. 2019, 15, 1984–1995, doi:10.3762/bjoc.15.194
- trifluoroacetate 60 by a known electrophilic substitution procedure that was developed by Tahara and co-workers [64]. Curtius rearrangement of 60 gave an isocyanate intermediate, which was reduced with LiAlH4 followed by reductive amination affording tertiary amine intermediate 61. Oxidation of 61 to its

Graphical Abstract

Figure 1: Structures of triptolide (1), triptonide (2), tripdiolide (3), 16-hydroxytriptolide (4), triptrioli...

Figure 2: Syntheses of triptolide.

Scheme 1: Berchtold’s synthesis of triptolide.

Scheme 2: Li’s formal synthesis of triptolide.

Scheme 3: van Tamelen’s asymmetric synthesis of triptonide and triptolide.

Scheme 4: Van Tamelen’s (method II) formal synthesis of triptolide.

Scheme 5: Sherburn’s formal synthesis of triptolide.

Scheme 6: van Tamelen’s biogenetic type total synthesis of triptolide.

Scheme 7: Yang’s total synthesis of triptolide.

Scheme 8: Key intermediates or transformations of routes J–N.
The design and synthesis of an antibacterial phenothiazine–siderophore conjugate
- Abed Tarapdar,
- James K. S. Norris,
- Oliver Sampson,
- Galina Mukamolova and
- James T. Hodgkinson
Beilstein J. Org. Chem. 2018, 14, 2646–2650, doi:10.3762/bjoc.14.242
- . In the final PMB global deprotection step we were pleased to observe the formation of our desired final phenothiazine–sideophore conjugate in moderate yield. The addition of anisole to the reaction mixture was found to be essential to inhibit competing electrophilic substitution side reactions. A

Graphical Abstract

Figure 1: NDH-2 is a validated target for 1 with an MIC of 1.1 µg/mL against M. tuberculosis.

Scheme 1: Synthesis of phenothiazine-PEG-amine component.

Scheme 2: Synthesis of the azotochelin siderophore component.

Scheme 3: Final conjugation and deprotection to yield a phenothiazine siderophore conjugate.
Applications of organocatalysed visible-light photoredox reactions for medicinal chemistry
- Michael K. Bogdos,
- Emmanuel Pinard and
- John A. Murphy
Beilstein J. Org. Chem. 2018, 14, 2035–2064, doi:10.3762/bjoc.14.179
- regioselective and follows the same substitution pattern as the electrophilic substitution of electron-rich heterocycles. Although highly elaborated structures are not presented, the mild reaction conditions and general functional group compatibility that the reaction exhibits make it well suited for LSF

Graphical Abstract

Figure 1: Depiction of the energy levels of a typical organic molecule and the photophysical processes it can...

Figure 2: General catalytic cycle of a photocatalyst in a photoredox organocatalysed reaction. [cat] – photoc...

Figure 3: Structures and names of the most common photocatalysts encountered in the reviewed literature.

Figure 4: General example of a reductive quenching catalytic cycle. [cat] – photocatalyst, [cat]* – photocata...

Figure 5: General example of an oxidative quenching catalytic cycle. [cat] – photocatalyst, [cat]* – photocat...

Scheme 1: Oxidative coupling of aldehydes and amines to amides using acridinium salt photocatalysis.

Figure 6: Biologically active molecules containing a benzamide linkage.

Scheme 2: The photocatalytic reduction of amino acids to produce the corresponding free or protected amines.

Scheme 3: The organocatalysed photoredox base-mediated oxidation of thiols to disulfides.

Scheme 4: C-Terminal modification of peptides and proteins using organophotoredox catalysis.

Scheme 5: The reduction and aryl coupling of aryl halides using a doubly excited photocatalyst (PDI).

Figure 7: Mechanism for the coupling of aryl halides using PDI, which is excited sequentially by two photons.

Scheme 6: The arylation of five-membered heteroarenes using arenediazonium salts under organophotoredox condi...

Scheme 7: The C–H (hetero)arylation of five-membered heterocycles under Eosin Y photocatalysis.

Scheme 8: The C–H sulfurisation of imidazoheterocycles using Eosin B-catalyzed photochemical methods.

Scheme 9: The introduction of the thiocyanate group using Eosin Y photocatalysis.

Scheme 10: Sulfonamidation of pyrroles using oxygen as the terminal oxidant.

Scheme 11: DDQ-catalysed C–H amination of arenes and heteroarenes.

Scheme 12: Photoredox-promoted radical Michael addition reactions of allylic or benzylic carbons.

Figure 8: Proposed mechanistic rationale for the observed chemoselectivities.

Scheme 13: The photocatalytic manipulation of C–H bonds adjacent to amine groups.

Scheme 14: The perylene-catalysed organophotoredox tandem difluoromethylation–acetamidation of styrene-type al...

Figure 9: Examples of biologically active molecules containing highly functionalised five membered heterocycl...

Scheme 15: The [3 + 2]-cycloaddition leading to the formation of pyrroles, through the reaction of 2H-azirines...

Figure 10: Proposed intermediate that determines the regioselectivity of the reaction.

Figure 11: Comparison of possible pathways of reaction and various intermediates involved.

Scheme 16: The acridinium salt-catalysed formation of oxazoles from aldehydes and 2H-azirines.

Scheme 17: The synthesis of oxazolines and thiazolines from amides and thioamides using organocatalysed photor...

Figure 12: Biologically active molecules on the market containing 1,3,4-oxadiazole moieties.

Scheme 18: The synthesis of 1,3,4-oxadiazoles from aldehyde semicarbazones using Eosin Y organophotocatalysis.

Scheme 19: The dimerization of primary thioamides to 1,2,4-thiadiazoles catalysed by the presence of Eosin Y a...

Scheme 20: The radical cycloaddition of o-methylthioarenediazonium salts and substituted alkynes towards the f...

Scheme 21: The dehydrogenative cascade reaction for the synthesis of 5,6-benzofused heterocyclic systems.

Figure 13: Trifluoromethylated version of compounds which have known biological activities.

Scheme 22: Eosin Y-catalysed photoredox formation of 3-substituted benzimidazoles.

Scheme 23: Oxidation of dihydropyrimidines by atmospheric oxygen using photoredox catalysis.

Scheme 24: Photoredox-organocatalysed transformation of 2-substituted phenolic imines to benzoxazoles.

Scheme 25: Visible light-driven oxidative annulation of arylamidines.

Scheme 26: Methylene blue-photocatalysed direct C–H trifluoromethylation of heterocycles.

Scheme 27: Photoredox hydrotrifluoromethylation of terminal alkenes and alkynes.

Scheme 28: Trifluoromethylation and perfluoroalkylation of aromatics and heteroaromatics.

Scheme 29: The cooperative asymmetric and photoredox catalysis towards the functionalisation of α-amino sp3 C–...

Scheme 30: Organophotoredox-catalysed direct C–H amidation of aromatics.

Scheme 31: Direct C–H alkylation of heterocycles using BF3K salts. CFL – compact fluorescent lamp.

Figure 14: The modification of camptothecin, demonstrating the use of the Molander protocol in LSF.

Scheme 32: Direct C–H amination of aromatics using acridinium salts.

Scheme 33: Photoredox-catalysed nucleophilic aromatic substitution of nucleophiles onto methoxybenzene derivat...

Scheme 34: The direct C–H cyanation of aromatics with a focus on its use for LSF.
Thiocarbonyl-enabled ferrocene C–H nitrogenation by cobalt(III) catalysis: thermal and mechanochemical
- Santhivardhana Reddy Yetra,
- Zhigao Shen,
- Hui Wang and
- Lutz Ackermann
Beilstein J. Org. Chem. 2018, 14, 1546–1553, doi:10.3762/bjoc.14.131
- preferentially, which can be rationalized with a base-assisted internal electrophilic substitution (BIES) [24][105] C–H cobaltation mechanism. In addition, the electron-rich amidating reagent 2c was found to be inherently more reactive (Scheme 5). As to further late-stage manipulation of the thus-obtained

Graphical Abstract

Figure 1: Selected ferrocene-based ligands and organocatalysts.

Scheme 1: Scope of substituted dioxazolones 2.

Scheme 2: C–H Amidation of arylated ferrocenes 1.

Scheme 3: Thiocarbonyl-assisted C–H amidation.

Scheme 4: H/D Exchange reactions.

Scheme 5: Intermolecular competition experiments.

Scheme 6: Synthesis of aminoketone 4aa.

Scheme 7: Mechanochemical ferrocene C–H nitrogenation.
One hundred years of benzotropone chemistry
- Arif Dastan,
- Haydar Kilic and
- Nurullah Saracoglu
Beilstein J. Org. Chem. 2018, 14, 1120–1180, doi:10.3762/bjoc.14.98

Graphical Abstract

Scheme 1: Tropone (1), tropolone (2) and their resonance structures.

Figure 1: Natural products containing a tropone nucleus.

Figure 2: Possible isomers 11–13 of benzotropone.

Scheme 2: Synthesis of benzotropones 11 and 12.

Scheme 3: Oxidation products of benzotropylium fluoroborate (16).

Scheme 4: Oxidation of 7-bromo-5H-benzo[7]annulene (22).

Scheme 5: Synthesis of 4,5-benzotropone (11) using o-phthalaldehyde (27).

Scheme 6: Synthesis of 4,5-benzotropone (11) starting from oxobenzonorbornadiene 31.

Scheme 7: Acid-catalyzed cleavage of oxo-bridge of 34.

Scheme 8: Synthesis of 4,5-benzotropone (11) from o-xylylene dibromide (38).

Scheme 9: Synthesis of 4,5-benzotropone (11) via the carbene adduct 41.

Scheme 10: Heck coupling strategy for the synthesis of 11.

Scheme 11: Synthesis of benzofulvalenes via carbonyl group of 4,5-benzotropone (11).

Figure 3: Some cycloheptatrienylium cations.

Scheme 12: Synthesis of condensation product 63 and its subsequent oxidative cyclization products.

Figure 4: A novel series of benzo[7]annulenes prepared from 4,5-benzotropone (11).

Scheme 13: Preparation of substituted benzo[7]annulene 72 using the Mukaiyama-Michael reaction.

Figure 5: Possible benzo[7]annulenylidenes 73–75.

Scheme 14: Thermal and photochemical decomposition of 7-diazo-7H-benzo[7]annulene (76) and the trapping of int...

Scheme 15: Synthesis of benzoheptafulvalene 86.

Scheme 16: Synthesis of 7-(diphenylmethylene)-7H-benzo[7]annulene (89).

Scheme 17: Reaction of 4,5-benzotropone (11) with dimethyl diazomethane.

Scheme 18: Synthesis of dihydrobenzomethoxyazocine 103.

Scheme 19: Synthesis and reducibility of benzo-homo-2-methoxyazocines.

Scheme 20: Synthesis of 4,5-benzohomotropones 104 and 115 from 4,5-benzotropones 11 and 113.

Scheme 21: A catalytic deuterogenation of 4,5-benzotropone (11) and synthesis of 5-monosubstituted benzo[7]ann...

Scheme 22: Synthesis of methyl benzo[7]annulenes 131 and 132.

Scheme 23: Ambident reactivity of halobenzo[7]annulenylium cations 133a/b.

Scheme 24: Preparation of benzo[7]annulenylidene–iron complexes 147.

Scheme 25: Synthesis of 1-ethynylbenzotropone (150) and the etheric compound 152 from 4,5-benzotropone (11) wi...

Scheme 26: Thermal decomposition of 4,5-benzotropone (11).

Scheme 27: Reaction of 4,5-benzotropone (11) with 1,2-ethanediol and 1,2-ethanedithiol.

Scheme 28: Conversions of 1-benzosuberone (162) to 2,3-benzotropone (12).

Scheme 29: Synthesis strategies for 2,3-bezotropone (12) using 1-benzosuberones.

Scheme 30: Oxidation-based synthesis of 2,3-benzotropone (12) via 1-benzosuberone (162).

Scheme 31: Synthesis of 2,3-benzotropone (12) from α-tetralone (171) via ring-expansion.

Scheme 32: Preparation of 2,3-benzotropone (12) by using of benzotropolone 174.

Figure 6: Benzoheptafulvenes as condensation products of 2,3-benzotropone (12).

Scheme 33: Conversion of 2,3-benzotropone (12) to tosylhydrazone salt 182 and gem-dichloride 187.

Figure 7: Benzohomoazocines 191–193 and benzoazocines 194–197.

Scheme 34: From 2,3-benzotropone (12) to carbonium ions 198–201.

Scheme 35: Cycloaddition reactions of 2,3-benzotropone (12).

Scheme 36: Reaction of 2,3-benzotropone (12) with various reagents and compounds.

Figure 8: 3,4-Benzotropone (13) and its resonance structure.

Scheme 37: Synthesis of 6,7-benzobicyclo[3.2.0]hepta-3,6-dien-2-one (230).

Figure 9: Photolysis and thermolysis products of 230.

Figure 10: Benzotropolones and their tautomeric structures.

Scheme 38: Synthesis strategies of 4,5-benzotropolone (238).

Scheme 39: Synthesis protocol for 2-hydroxy-4,5-benzotropone (238) using oxazole-benzo[7]annulene 247.

Figure 11: Some quinoxaline and pyrazine derivatives 254–256 prepared from 4,5-benzotropolone (238).

Scheme 40: Nitration product of 4,5-benzotropolone (238) and its isomerization to 1-nitro-naphthoic acid (259)....

Scheme 41: Synthesis protocol for 6-hydroxy-2,3-benzotropone (239) from benzosuberone (162).

Scheme 42: Various reactions via 6-hydroxy-2,3-benzotropone (239).

Scheme 43: Photoreaction of 6-hydroxy-2,3-benzotropone (239).

Scheme 44: Synthesis of 7-hydroxy-2,3-benzotropone (241) from benzosuberone (162).

Scheme 45: Synthesis strategy for 7-hydroxy-2,3-benzotropone (241) from ketone 276.

Scheme 46: Synthesis of 7-hydroxy-2,3-benzotropone (241) from β-naphthoquinone (280).

Scheme 47: Synthesis of 7-hydroxy-2,3-benzotropone (241) from bicyclic endoperoxide 213.

Scheme 48: Synthesis of 7-hydroxy-2,3-benzotropone (241) by ring-closing metathesis.

Figure 12: Various monosubstitution products 289–291 of 7-hydroxy-2,3-benzotropone (241).

Scheme 49: Reaction of 7-hydroxy-2,3-benzotropone (241) with various reagents.

Scheme 50: Synthesis of 4-hydroxy-2,3-benzotropones 174 and 304 from diketones 300/301.

Scheme 51: Catalytic hydrogenation of diketones 300 and 174.

Scheme 52: Synthesis of halo-benzotropones from alkoxy-naphthalenes 306, 307 and 310.

Figure 13: Unexpected byproducts 313–315 during synthesis of chlorobenzotropone 309.

Figure 14: Some halobenzotropones and their cycloadducts.

Scheme 53: Multisep synthesis of 2-chlorobenzotropone 309.

Scheme 54: A multistep synthesis of 2-bromo-benzotropone 26.

Scheme 55: A multistep synthesis of bromo-2,3-benzotropones 311 and 316.

Scheme 56: Oxidation reactions of 8-bromo-5H-benzo[7]annulene (329) with some oxidants.

Scheme 57: Synthesis of 2-bromo-4,5-benzotropone (26).

Scheme 58: Synthesis of 6-chloro-2,3-benzotropone (335) using LiCl and proposed intermediate 336.

Scheme 59: Reaction of 7-bromo-2,3-benzotropone (316) with methylamine.

Scheme 60: Reactions of bromo-2,3-benzotropones 26 and 311 with dimethylamine.

Scheme 61: Reactions of bromobenzotropones 311 and 26 with NaOMe.

Scheme 62: Reactions of bromobenzotropones 26 and 312 with t-BuOK in the presence of DPIBF.

Scheme 63: Cobalt-catalyzed reductive cross-couplings of 7-bromo-2,3-benzotropone (316) with cyclic α-bromo en...

Figure 15: Cycloadduct 357 and its di-π-methane rearrangement product 358.

Scheme 64: Catalytic hydrogenation of 2-chloro-4,5-benzotropone (311).

Scheme 65: Synthesis of dibromo-benzotropones from benzotropones.

Scheme 66: Bromination/dehydrobromination of benzosuberone (162).

Scheme 67: Some transformations of isomeric dibromo-benzotropones 261A/B.

Scheme 68: Transformations of benzotropolone 239B to halobenzotropolones 369–371.

Figure 16: Bromobenzotropolones 372–376 and 290 prepared via bromination/dehydrobromination strategy.

Scheme 69: Synthesis of some halobenzotropolones 289, 377 and 378.

Figure 17: Bromo-chloro-derivatives 379–381 prepared via chlorination.

Scheme 70: Synthesis of 7-iodo-3,4-benzotropolone (382).

Scheme 71: Hydrogenation of bromobenzotropolones 369 and 370.

Scheme 72: Debromination reactions of mono- and dibromides 290 and 375.

Figure 18: Nitratation and oxidation products of some halobenzotropolenes.

Scheme 73: Azo-coupling reactions of some halobenzotropolones 294, 375 and 378.

Figure 19: Four possible isomers of dibenzotropones 396–399.

Figure 20: Resonance structures of tribenzotropone (400).

Scheme 74: Two synthetic pathways for tribenzotropone (400).

Scheme 75: Synthesis of tribenzotropone (400) from dibenzotropone 399.

Scheme 76: Synthesis of tribenzotropone (400) from 9,10-phenanthraquinone (406).

Scheme 77: Synthesis of tribenzotropone (400) from trifluoromethyl-substituted arene 411.

Figure 21: Dibenzosuberone (414).

Figure 22: Reduction products 415 and 416 of tribenzotropone (400).

Figure 23: Structures of tribenzotropone dimethyl ketal 417 and 4-phenylfluorenone (412) and proposed intermed...

Figure 24: Structures of benzylidene- and methylene-9H-tribenzo[a,c,e][7]annulenes 419 and 420 and chiral phos...

Figure 25: Structures of tetracyclic alcohol 422, p-quinone methide 423 and cation 424.

Figure 26: Structures of host molecules 425–427.

Scheme 78: Synthesis of non-helical overcrowded derivatives syn/anti-431.

Figure 27: Hexabenzooctalene 432.

Figure 28: Structures of possible eight isomers 433–440 of naphthotropone.

Scheme 79: Synthesis of naphthotropone 437 starting from 1-phenylcycloheptene (441).

Scheme 80: Synthesis of 10-hydroxy-11H-cyclohepta[a]naphthalen-11-one (448) from diester 445.

Scheme 81: Synthesis of naphthotropone 433.

Scheme 82: Synthesis of naphthotropones 433 and 434 via cycloaddition reaction.

Scheme 83: Synthesis of naphthotropone 434 starting from 452.

Figure 29: Structures of tricarbonyl(tropone)irons 458, and possible cycloadducts 459.

Scheme 84: Synthesis of naphthotropone 436.

Scheme 85: Synthesis of precursor 465 for naphthotropone 435.

Scheme 86: Generation of naphthotropone 435 from 465.

Figure 30: Structures of tropylium cations 469 and 470.

Figure 31: Structures of tropylium ions 471+.BF4−, 472+.BF4−, and 473+.BF4−.

Scheme 87: Synthesis of tropylium ions 471+.BF4− and 479+.ClO4−.

Scheme 88: Synthesis of 1- and 2-methylanthracene (481 and 482) via carbene–carbene rearrangement.

Figure 32: Trapping products 488–490.

Scheme 89: Generation and chemistry of a naphthoannelated cycloheptatrienylidene-cycloheptatetraene intermedia...

Scheme 90: Proposed intermediates and reaction pathways for adduct 498.

Scheme 91: Exited-state intramolecular proton transfer of 505.

Figure 33: Benzoditropones 506 and 507.

Scheme 92: Synthesis of benzoditropone 506e.

Scheme 93: Synthetic approaches for dibenzotropone 507 via tropone (1).

Scheme 94: Formation mechanisms of benzoditropone 507 and 516 via 515.

Scheme 95: Synthesis of benzoditropones 525 and 526 from pyromellitic dianhydride (527).

Figure 34: Possible three benzocyclobutatropones 534–536.

Scheme 96: Synthesis of benzocyclobutatropones 534 and 539.

Scheme 97: Synthesis attempts for benzocyclobutatropone 545.

Scheme 98: Generation and trapping of symmetric benzocyclobutatropone 536.

Scheme 99: Synthesis of chloro-benzocyclobutatropone 552 and proposed mechanism of fluorenone derivatives.

Scheme 100: Synthesis of tropolone analogue 559.

Scheme 101: Synthesis of tropolones 561 and 562.

Figure 35: o/p-Tropoquinone rings (563 and 564) and benzotropoquinones (565–567).

Scheme 102: Synthesis of benzotropoquinone 566.

Scheme 103: Synthesis of benzotropoquinone 567 via a Diels–Alder reaction.

Figure 36: Products 575–577 through 1,2,3-benzotropoquinone hydrate 569.

Scheme 104: Structures 578–582 prepared from tropoquinone 567.

Figure 37: Two possible structures 583 and 584 for dibenzotropoquinone, and precursor compound 585 for 583.

Scheme 105: Synthesis of saddle-shaped ketone 592 using dibenzotropoquinone 584.






